Functional OCT of retinal neurovascular coupling
Retinal neurodegenerative diseases, including age-related macular degeneration (AMD), retinitis pigmentosa (RP), diabetic retinopathy (DR), and glaucoma, can cause significant vision loss without timely medical interventions. Moreover, as part of the central nervous system (CNS), the retina is also affected by other neurodegenerative diseases, such as Parkinson’s and Alzheimer’s diseases, which are the primary causes of dementia. Early detection of these conditions is crucial for studying and developing preventive strategies. Functional imaging of neurovascular coupling defects promises early detection of neurodegeneration, but direct access to the brain for high-resolution examination is challenging. The retina provides a unique window for high-resolution study of neurovascular coupling defects. Recent advances in functional optical coherence tomography (OCT) have enabled imaging of transient intrinsic optical signal (IOS) changes correlated with stimulus-activated retinal neural activity and hemodynamic response. Functional OCT of neurovascular coupling holds the potential for high spatiotemporal resolution methodology to investigate coherent interactions between neural activities and hemodynamic changes in the retina, offering a promising tool for early detection and better management of neurodegenerative diseases.
Figure Heading link
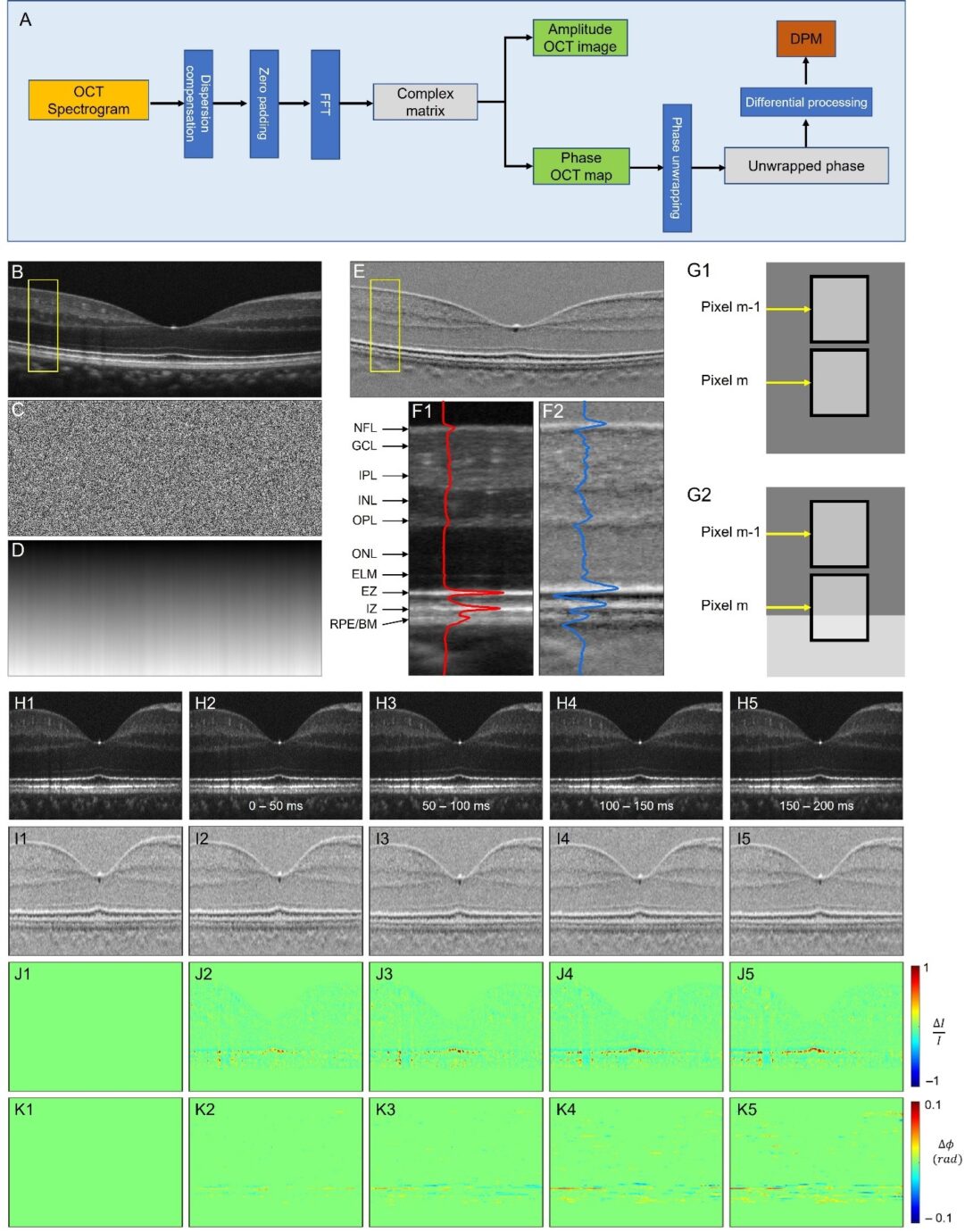
Fig. 1. Differential phase mapping (DPM) and DPM IOS. (A) A flow chart of amplitude OCT and differential phase mapping (DPM) processing. Representative OCT amplitude image (B), corresponding phase map (C), unwrapped phase map (D), and DPM (E). (F) Flattened amplitude (F1) and DPM (F2) images corresponding to the yellow rectangular areas in (B) and (E). The blue and red curves show the waveform profiles of the amplitude image and DPM. (G) Schematic diagram of differential phase interpretation. (H) Amplitude image sequence. (I) DPM sequence. (J) Amplitude-IOS sequence. (K) Phase-IOS sequence. For more details, please see here.
Current development and future direction Heading link
Functional imaging of animal models
Our team, under the guidance of Dr. Xincheng Yao, has been extensively involved in functional IOS imaging since 2005. Our research has included the observation of the IOS from the outer retina (photoreceptor) to the inner retina (GCL, IPL, and OPL) in order to better understand the mechanisms involved in phototransduction. In addition to observing the IOS, we have also investigated the signal source of IOS from the outer retina using OCT and microscopy. Our research has aimed to identify the underlying mechanisms that generate IOS signals in order to develop IOS as a diagnostic biomarker to detect various retina diseases.
Based on our findings, we have attempted to demonstrate the feasibility of using IOS imaging to detect various retina diseases, including retina degeneration and Alzheimer’s Disease, using multiple disease mouse models. By using these models, we hope to identify potential biomarkers for early diagnosis and better understanding of the underlying mechanisms of these diseases. We also investigated hemodynamic IOS together with neural IOS for neurovascular coupling assessment.
Functional imaging of humans
The aim of human IOS imaging is to discover new biomarkers that enable early detection of retinal disease. In our study, we successfully demonstrated the feasibility of non-mydriatic IOS mapping of retinal photoreceptors in conscious human subjects. By using depth-resolved optical coherence tomography, we were able to verify the anatomic origin of the fast photoreceptor-IOS and understand the mechanism of the rapid photoreceptor-IOS response. Furthermore, our study showed that it is possible to conduct simultaneous optical coherence tomography of amplitude-IOS and phase-IOS changes in human photoreceptors, which can be attributed to transient changes in outer segment morphology associated with phototransduction activation.
Future directions
Our research has laid a solid foundation for further exploration and development of neural and hemodynamic IOS imaging techniques for both animal and human study, which has the potential to revolutionize the diagnosis and treatment of retina diseases. We are seeking to expand our research to develop a novel functional ophthalmic imaging technology.
Related publications Heading link
2024
- Taeyoon Son, Guangying Ma, and Xincheng Yao, “Functional OCT reveals anisotropic changes of retinal flicker-evoked vasodilation,” Opt. Lett. 49, 2121-2124 (2024).[Full text]
- Ding Jie, Tae-Hoon Kim, Guangying Ma and Xincheng Yao (2024) “Intrinsic signal optoretinography of dark adaptation abnormality due to rod photoreceptor degeneration.” Exp. Biol. Med. 249:10024. doi: 10.3389/ebm.2024.10024.[Full text]
2023
- Adejumo, Tobiloba , Guangying Ma, Taeyoon Son, Tae-Hoon Kim, David Le, Albert K Dadzie, Shaiban Ahmed, and Xincheng Yao, “Adaptive vessel tracing and segmentation in OCT enables the robust detection of wall-to-lumen ratio abnormalities in 5xFAD mice,” Biomed. Opt. Express 14, 6350-6360 (2023).[Full text]
- Ma, Guangying, Tae-Hoon Kim, Taeyoon Son, Jie Ding, Shaiban Ahmed, Tobiloba Adejumo, and Xincheng Yao, “Intrinsic signal optoretinography revealing AD-induced retinal photoreceptor hyperexcitability before a detectable morphological abnormality,” Opt. Lett. 48, 5129-5132 (2023).[Full text]
- Ma, Guangying, Taeyoon Son, Tobiloba Adejumo, and Xincheng Yao. “Rotational Distortion and Compensation in Optical Coherence Tomography with Anisotropic Pixel Resolution.” Bioengineering 10, no. 3 (2023): 313. [Full text]
2022
- Kim, Tae-Hoon, Guangying Ma, Taeyoon Son, and Xincheng Yao. “Functional Optical Coherence Tomography for Intrinsic Signal Optoretinography: Recent Developments and Deployment Challenges.” Frontiers in Medicine (2022). [Full text]
- Kim, Tae-Hoon, Jie Ding, and Xincheng Yao. “Intrinsic signal optoretinography of dark adaptation kinetics.” Scientific Reports 12, no. 1 (2022): 1-11. [Full text]
- Ma, Guangying, Jie Ding, Tae-Hoon Kim, and Xincheng Yao. “Quantitative Optical Coherence Tomography for Longitudinal Monitoring of Postnatal Retinal Development in Developing Mouse Eyes.” Applied Sciences 12, no. 4 (2022): 1860. [Full text]
- Adejumo, Tobiloba, Tae-Hoon Kim, David Le, Taeyoon Son, Guangying Ma, and Xincheng Yao, “Depth-resolved vascular profile features for artery-vein classification in OCT and OCT angiography of human retina,” Biomed. Opt. Express 13, 1121-1130 (2022). [Full text]
2021
- Kim, Tae-Hoon, and Xincheng Yao. “Longitudinal optical coherence tomography and angiography of hyaloid vascular regression in developing mouse eyes.” Photo Acoustic and Optical Coherence Tomography Imaging, Volume 3 (2021). [Full text]
- Kim, Tae-Hoon, Taeyoon Son, Dieter Klatt, and Xincheng Yao. “Concurrent OCT and OCT angiography of retinal neurovascular degeneration in the 5XFAD Alzheimer’s disease mice.” Neurophotonics 8, no. 3 (2021): 035002. [Full text]
- Yao, Xincheng, Taeyoon Son, Tae-Hoon Kim, and David Le. “Interpretation of anatomic correlates of outer retinal bands in optical coherence tomography.” Experimental Biology and Medicine (2021): 15353702211022674. [Full text]
- Ma, Guangying, Taeyoon Son, Tae-Hoon Kim, and Xincheng Yao. Functional optoretinography: concurrent OCT monitoring of intrinsic signal amplitude and phase dynamics in human photoreceptors. Biomedical optics express 2021;12:2661-69. [Full text]
- Son, Taeyoon, Tae-Hoon Kim, Guangying Ma, Hoonsup Kim, and Xincheng Yao. Functional intrinsic optical signal imaging for objective optoretinography of human photoreceptors. Exp Biol Med (Maywood) 2021;246:639-43 [Full text]
- Ma, Guangying , Taeyoon Son, Tae-Hoon Kim, and Xincheng Yao. “In vivo optoretinography of phototransduction activation and energy metabolism in retinal photoreceptors.” Journal of Biophotonics (2021): e202000462. [Full text]
- Kim, Tae-Hoon, David Le, Taeyoon Son, and Xincheng Yao, “Vascular morphology and blood flow signatures for differential artery-vein analysis in optical coherence tomography of the retina” Biomed. Opt. Express 12, 367-379 (2021) [Full text]
2020
- Kim, Tae-Hoon, Benquan Wang, Yiming Lu, Taeyoon Son, and Xincheng Yao, “Functional optical coherence tomography enables in vivo optoretinography of photoreceptor dysfunction due to retinal degeneration,” Biomed. Opt. Express 11, 5306-5320 (2020) [Full text]
- Yao, Xincheng, and Tae-Hoon Kim. “Fast Intrinsic Optical Signal Correlates with Activation Phase of Phototransduction in Retinal Photoreceptors.” Experimental Biology and Medicine, June 2020 [Full text]
- Lu, Yiming, Tae-Hoon Kim, and Xincheng Yao. “Comparative Study of Wild-Type and Rd10 Mice Reveals Transient Intrinsic Optical Signal Response before Phosphodiesterase Activation in Retinal Photoreceptors.” Experimental Biology and Medicine 245, no. 4 (February 2020): 360–67. [Full text]
- Yao, Xincheng, Minhaj Nur Alam, David Le, and Devrim Toslak. “Quantitative Optical Coherence Tomography Angiography: A Review.” Experimental Biology and Medicine, (January 2020). [Full text]
2019
- Kim, Tae-Hoon, Taeyoon Son, David Le, Xincheng Yao. “Longitudinal OCT and OCTA monitoring reveals accelerated regression of hyaloid vessels in retinal degeneration 10 (rd10) mice.” Sci Rep 9, 16685 (2019) doi:10.1038/s41598-019-53082-9 [Full text].
- Kim, Tae-Hoon, Taeyoon Son, and Xincheng Yao. “Functional OCT angiography reveals early physiological dysfunction of hyaloid vasculature in developing mouse eye.” Experimental Biology and Medicine 244, no. 10 (2019): 819-823. [Full text]
- Son, Taeyoon, Minhaj Alam, Tae-Hoon Kim, Changgeng Liu, Devrim Toslak, and Xincheng Yao. “Near infrared oximetry-guided artery–vein classification in optical coherence tomography angiography.” Experimental Biology and Medicine 244, no. 10 (2019): 813-818. [Full text]
2018
- Yao, Xincheng, Taeyoon Son, Tae-Hoon Kim, and Yiming Lu. “Functional optical coherence tomography of retinal photoreceptors.” Experimental Biology and Medicine 243, no. 17-18 (2018): 1256-1264. [Full Text]
- Lu, Yiming, Jacopo Benedetti, and Xincheng Yao. “Light-induced length shrinkage of rod photoreceptor outer segments.” Translational Vision Science & Technology 7, no. 6 (2018): 29-29. [Full Text]
- Kim, Tae-Hoon, Taeyoon Son, Yiming Lu, Minhaj Alam, and Xincheng Yao. “Comparative optical coherence tomography angiography of wild-type and rd10 mouse retinas.” Translational vision science & technology 7, no. 6 (2018): 42-42. [Full Text]
- Son, Taeyoon, Minhaj Alam, Devrim Toslak, Benquan Wang, Yiming Lu, and Xincheng Yao. “Functional optical coherence tomography of neurovascular coupling interactions in the retina.” Journal of biophotonics 11, no. 12 (2018): e201800089. [Full Text]
- Lu, Yiming, Changgeng Liu, and Xincheng Yao. “In vivo super-resolution imaging of transient retinal phototropism evoked by oblique light stimulation.” Journal of biomedical optics 23, no. 5 (2018): 050502-050502.[Full Text]
2017
- Thapa, Damber, Benquan Wang, Yiming Lu, Taeyoon Son, and Xincheng Yao. “Enhancement of intrinsic optical signal recording with split spectrum optical coherence tomography.” Journal of modern optics 64, no. 17 (2017): 1800-1807. [Full Text]
- Lu, Yiming, Benquan Wang, David R. Pepperberg, and Xincheng Yao. “Stimulus-evoked outer segment changes occur before the hyperpolarization of retinal photoreceptors.” Biomedical optics express 8, no. 1 (2017): 38-47. [Full Text]
2016
- Wang, Benquan , Yiming Lu, and Xincheng Yao. “In vivo optical coherence tomography of stimulus-evoked intrinsic optical signals in mouse retinas.” Journal of biomedical optics 21, no. 9 (2016): 096010-096010. [Full Text]
- Son, Taeyoon, Benquan Wang, Damber Thapa, Yiming Lu, Yanjun Chen, Dingcai Cao, and Xincheng Yao. “Optical coherence tomography angiography of stimulus evoked hemodynamic responses in individual retinal layers.” Biomedical optics express 7, no. 8 (2016): 3151-3162.[Full Text]
- Zhao, Xiaohui, Damber Thapa, Benquan Wang, Yiming Lu, Shaoyan Gai, and Xincheng Yao. “Stimulus-evoked outer segment changes in rod photoreceptors.” Journal of biomedical optics 21, no. 6 (2016): 065006-065006. [Full Text]
2015
- Yao, Xincheng, and Benquan Wang. “Intrinsic optical signal imaging of retinal physiology: a review.” Journal of biomedical optics 20, no. 9 (2015): 090901-090901. [Full Text].
- Zhang, Qiuxiang, Rongwen Lu, Benquan Wang, Jeffrey D. Messinger, Christine A. Curcio, and Xincheng Yao. “Functional optical coherence tomography enables in vivo physiological assessment of retinal rod and cone photoreceptors.” Scientific reports 5, no. 1 (2015): 9595. [Full Text]





